- l'étude des mécanismes corticaux qui induisent ou qui modulent la perception de la douleur chez l'Homme (Physiologie de la Douleur) ;
- l'utilisation de techniques neurophysiologiques pour améliorer notre compréhension, le diagnostic et la prédiction de la douleur neuropathique (Patho-physiologie de la Douleur) ;
- l'amélioration des procédures de neurostimulation corticales pour diminuer la douleur par une meilleure compréhension de leurs mécanismes (NeuroStimulation).
L'équipe est dans l'hôpital Neurologique de Lyon et dans la faculté de médecine de l'université de Saint-Etienne. Tous les chercheurs de l'équipe ont des activités cliniques ce qui permet d'avoir un accès direct aux équipements hospitaliers et à la patientèle.
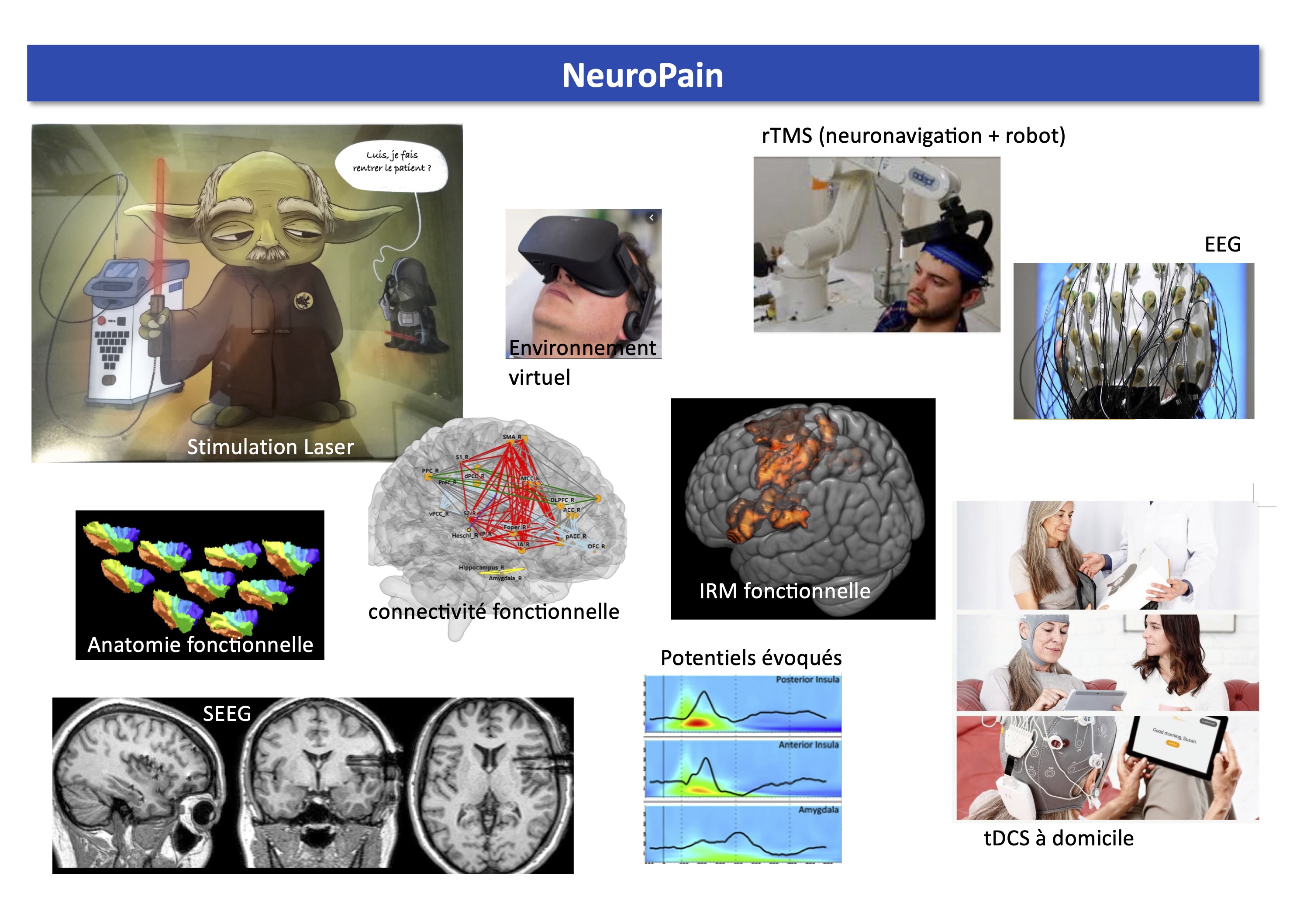
NeuroPain a accès à des systèmes de stimulation corticale (rTMS, tDCS, stimulation épidurale et intracorticale). Nos systèmes rTMS permettent de localiser précisément la cible grâce à la neuronavigation 3D à partir d'une image anatomique IRM. De plus, le sytème au CHU de Saint-Etienne a un bras robotisé qui permet le maintien de la sonde à sa meilleure position malgré le mouvement de la tête du sujet. Nous avons aussi accès à divers systèmes de stimulations sensorielles comme un thermotest et des lasers Nd:YAP.
Nous utilisons divers types d'enregistrement des activités cérébrales : électrophysiologie à haute densité (EEG 2x128 canaux), MEG, IRM 3T, TEP, EEG intracrânienne. Enfin, NeuroPain est un membre fondateur de la plateforme "Neuro-Immersion" qui couple la réalité virtuelle avec de l'EEG-HD, de la TMS et de la cinématique en 3D et en temps réel.
EEG = électroencéphalographie
rTMS = stimulation magnétique transcrânienne répétée
tDCS = stimulation
TEP = tomographie par émission de positron
IRM = imagerie par résonance magnétique
MEG = magnétoencéphalographie

- 1 chercheuse INSERM et 1 MC de l'université de St-Etienne
- 2 ingénieures
- 14 cliniciens (Neurologues, Neurochirurgiens et Psychiatres)
Psychophysique, EEG, sEEG intracrânienne, IRM, TEP, rTMS avec neuro-navigation, tDCS, environnement virtuel
Physiologie et modulation de la douleur
• Analyse spatio-temporelle du traitement de l’information douloureuse
- Compréhension des déterminants corticaux de la perception subjective de la douleur
(M. Frot, M. Guenot , H. Bastuji)
- Analyses des interactions fonctionnelles intra-cérébrales en lien avec la perception douloureuse
(C. Fauchon, L. Modernell, I. Faillenot)
• Interaction avec des mécanismes cognitifs attentionnels et émotionnels
- Intégration corticale des visages émotionnels douloureux
(M. Frot)
- Décodage des signaux douloureux dans un stimulus auditif, le cri du bébé
(S. Corvin, C. Fauchon, I. Faillenot, R. Peyron, Coll N. Mathevon, ENES)
• Mémorisation de la douleur
(M. Frot, C. Perchet)
- Mémorisation de la douleur en lien avec un contexte
- Mémorisation du contexte associé à la douleur
Diagnostic, prévention et caractérisation de la douleur neuropathique
• Projet européen sur la détection des biomarqueurs de la douleur
(L. Garcia-Larrea)
• Potentiels Évoqués Laser en clinique pour le diagnostic des douleurs neuropathiques
(L. Garcia-Larrea, C. Perchet)
Traitements non médicamenteux de la douleur chronique
• Etude de la rTMS et tDCS à visée antalgique
- Suivi d’une cohorte de patients traités par rTMS puis par électrode corticale
(R. Peyron, J. Thomas, C. Quesada, L. Garcia-Larrea, F. Vassal)
- Programme de stimulation électrique transcrânienne (tDCS) à domicile (Projet STIMADOM)
(C. Salameh, C. Perchet, L. Garcia-Larrea)
• Couplage de la rTMS à la réalité virtuelle immersive
- Optimiser les effets de la rTMS en la couplant avec la thérapie miroir (Projet STIRM)
(S. Grenouillet, J. Thomas, C. Quesada, R. Peyron)
- Connectivité fonctionnelle corticale associée à l’efficacité du traitement
(L. Malagutti-Modernell, C. Fauchon, I. Faillenot, R. Peyron)
• Effet croisé de la rTMS des patients douloureux et dépressifs
cible corticale de stimulation : moteur primaire ou le préfrontal dorsolatéral (Projet DoloDep)
(N. Oriol, S. Grenouillet, I. Faillenot, R. Peyron)
• Efficacité de la rTMS du cortex moteur dans les douleurs neuropathiques du cancer (Projet NeuroCanPain)
(S. Grenouillet, C. Fauchon, R. Peyron)
• Neurostimulation électrique transcrânienne personalisée à visée antalgique (Projet PersoStim)
(C. Salameh, N. André-Obadia, L. Garcia-Larrea)
• Apport de la danse-thérapie chez des adolescents (Projet AlgoDanse)
(A. Bregeon, M. Frot, C. Perchet, G. Demarquay en collaboration avec le
Centre d’Etude de la Douleur de l’Hôpital Neurologique)
- Researchers
Philippe Convers
Jean Isnard
Amandine Rey
Michel Magnin
- Post-Doc or associate researchers
Juliette Gélébart (post-Doc, France)
Siloé Corvin (post-Doc, France)
Camille Fauchon (post-Doc, France)
Koichi Hagiwara (associated researcher, Japan) 2015-2019
Philip Jackson (Visiting Professor, Canada) 2013
Florian Chouchou (post-Doc, France)
Benjamin Dominguez-Trejo (Visiting Professor, Mexico)
Emile Simon (associate researcher)
Agustina Lascano (visiting neurologist, Suisse)
Stéphanie Maza (associate researcher, France)
Fabio Godinho (Post-Doc, Brazil)
Nuuti Vartiainen (Post-Doc, FInland)
Florian Chouchou (Post-Doc, France)
Sigrid Shuh-Hoffer (Post-Doc, Germany)
Valery Legrain (Post-Doc, Belgium)
Ron Kuppers (Post-Doc, Belgium)
Blanca Diaz (Post-Doc, Spain)
Jordi Pedro (Post-Doc, Spain)
Lorena Guzman (Post-Doc, Venezuela)
Aline Acosta (Post-Doc, Brazil)
Cecilia Flores (Post-Doc, Spain)
- PhD
Louis Tremblais (PhD, Ingénierie & santé, Saint-Etienne)
Juliette Van Eycken (PhD, Neurosciences, Lyon)
Solène Grenouillet (PhD, Ingénierie & santé, Saint-Etienne)
Charbel Salameh (PhD, Neurosciences, Lyon)
Laura Malaguti-Modernell (PhD, Neurosciences, Lyon) 2025
Joy Thomas (PhD, Ingénierie & santé, Saint-Etienne) 2024
Siloé Corvin (PhD, Neurosciences, Lyon) 2023
Juliette Gélébart (PhD, Neurosciences, Lyon) 2022
Argitxu Caldichoury (PhD, Neurosciences, Lyon) 2022
Benjamin Pommier (PhD, Ingénierie & santé, Saint-Etienne) 2019
Charles Quesada (PhD, Ingénierie & santé, Saint-Etienne) 2018
Camille Fauchon (PhD, Ingénierie & santé, Saint-Etienne) 2017
Christophe Nuti (PhD, Ingénierie & santé, Saint-Etienne)
Claire Bradley (PhD, Neurosciences, Lyon)
Claire Czelkala (PhD, Neurosciences, Lyon) 2015
Lea Claude (PhD, Neurosciences, Lyon) 2015
Anaïs Chapon (PhD, Neurosciences, Lyon) 2016
Bérangère Houzé (PhD, Neurosciences, Lyon) 2011
Laure Mazolla (PhD, Ingénierie & santé, Saint-Etienne) 2011
Florence Borgetto Pomares (PhD, Neurosciences, Lyon) 2011
Estelle Raffin (PhD, Neurosciences, Saint-Etienne) 2011
Aurelia Poujois (PhD, Saint-Etienne) 2011
Florian Chouchou (PhD, Neurosciences, Lyon)
Laure Peter-Derex (PhD, Neurosciences, Lyon)
Joseph Maraawi (PhD)
Mustafa Mahmutoglu (visiting PhD, Germany)
Marcos-Fortunato-de-Barrios Jr (visiting PhD, Germany)
Marina Pidal (visiting PhD, Spain)
German Prados (visiting PhD, Spain)
- master out of Lyon or St-Etienne
Emie Just (Master Toulouse)
Clementine Pouliot (Visiting Master, Canada)
Marie-Hélène Tessier (Visiting Master, Canada)
Nyamh Joyce (Master Erasmus, UK)
Ruth Kirby (Master Erasmus, UK)
Temitope Lana (Master Erasmus, UK)
Eva Masson (Master, Bordeaux)
Elisa Redavide (Master Erasmus, Italy)
Inès Jani (Master, Canada)
Martin-Schulz (Visiting Master, Germany)
Quentin Welniarz (Master, ENS Paris)
Laura Lucarelli, gestionnaire de l'équipe NeuroPain
Isabelle Faillenot, webmaster de l'équipe NeuroPain











